Accurate and Easy Testing
AZOVA is an exclusive distributor of one of the ONLY FDA authorized and EUA approved self administered COVID-19 PCR Saliva tests. We've brought this exclusive opportunity to your pharmacy so your patients can also benefit. No more painful nasal swabs for patients. The at home COVID-19 Saliva test only needs a saliva sample and is highly accurate, With 98% sensitivity and 99% specificity. Start offering this test today and generate revenue in four ways.
Make $22 to $29 in test administration fees for each test that you administer in your pharmacy through the AZOVA Digital Pharmacy Clinical Services Network program
Because this is the only self-administered saliva test kit that you can offer right from your pharmacy, you can simply provide the kit and let the patient complete the testing process and mail it back to the lab or you can mail it back to the lab. Here is how it works:
- You'll receive a custom-branded COVID testing microsite where patients can choose an at home or in pharmacy test. Place a link to this site on your website, in your social media and text/email it out to your patients.
- You must purchase inventory for in pharmacy testing.
- Your pharmacy will be listed in the national COVID testing network for employers, schools and universities to send their patients for testing.
- Patients register online and pay $149 or in pharmacy testing or $168 for at home testing. Patients may also enter an eligibility code when their organization covers the cost of testing
- You will receive a notification on your AZOVA dashboard that the patient has registered.
- The patient arrives at the pharmacy and you verify that the patient did register online. You must send an electronic order to the lab and complete the patient's encounter in order to be reimbursed for your test administration services.
- Please note: WE’VE UPGRADED THE PROGRAM TO NOW OFFER NEXT DAY SHIPPING BOTH WAYS. Provide the test kit to the patient. The patient will complete the test and take it to the nearest UPS location or the patient can give it back to you for you to deliver to UPS. The kit comes with a pre-paid next day UPS shipping label on the box.
- Results are returned to you and the patient within 72 hours of the lab receiving the specimen.
- AZOVA will reimburse you for what you paid for each test kit that you administer when patients register through your microsite for an in pharmacy test plus and administration fee of $22 to $29. You will also receive a $5 marketing fee for each patient who purchases an at home COVID test from your website.
COVID testing custom website

National COVID Testing Network
Offer in home COVID-19 testing from your website. Get paid $5 per test ordered from your website. Requires no inventory. You don’t need to do anything but add a link to your website.
You can choose to offer only at home testing if you'd like.
- You will receive a custom branded microsite for your website. Add the link to your website and social media and email/text it to your patients.
- Patients register for at home testing and AZOVA will provide all lab ordering, shipping and will send the results to the patient.
On site events and mass testing events for employers, schools and universities.
Your membership in the Digital Pharmacy Clinical Services Network comes complete with onsite testing event management. Pharmacists across the country are successfully managing COVID testing services for schools, universities and employers in the community. You can contract directly with any organization to offer and manage their COVID testing. This is how it works:
- You create an onsite event registration page for each organization that you are offering testing for. Email this out to the organization ahead of time so each enrollee can register online before getting tested at the event.
- AZOVA provides the software needed to manage the event, for patient registration, electronic laboratory ordering and electronic results reporting. In addition, all positive results will be reported to appropriate state agencies.
- You must purchase the inventory for each event or the employer can purchase the inventory directly and AZOVA will ship it to the employer event where you manage the testing and lab ordering.
- AZOVA will send the test results to you and the patient electronically. Patients who need it can get a telemedicine visit as needed.
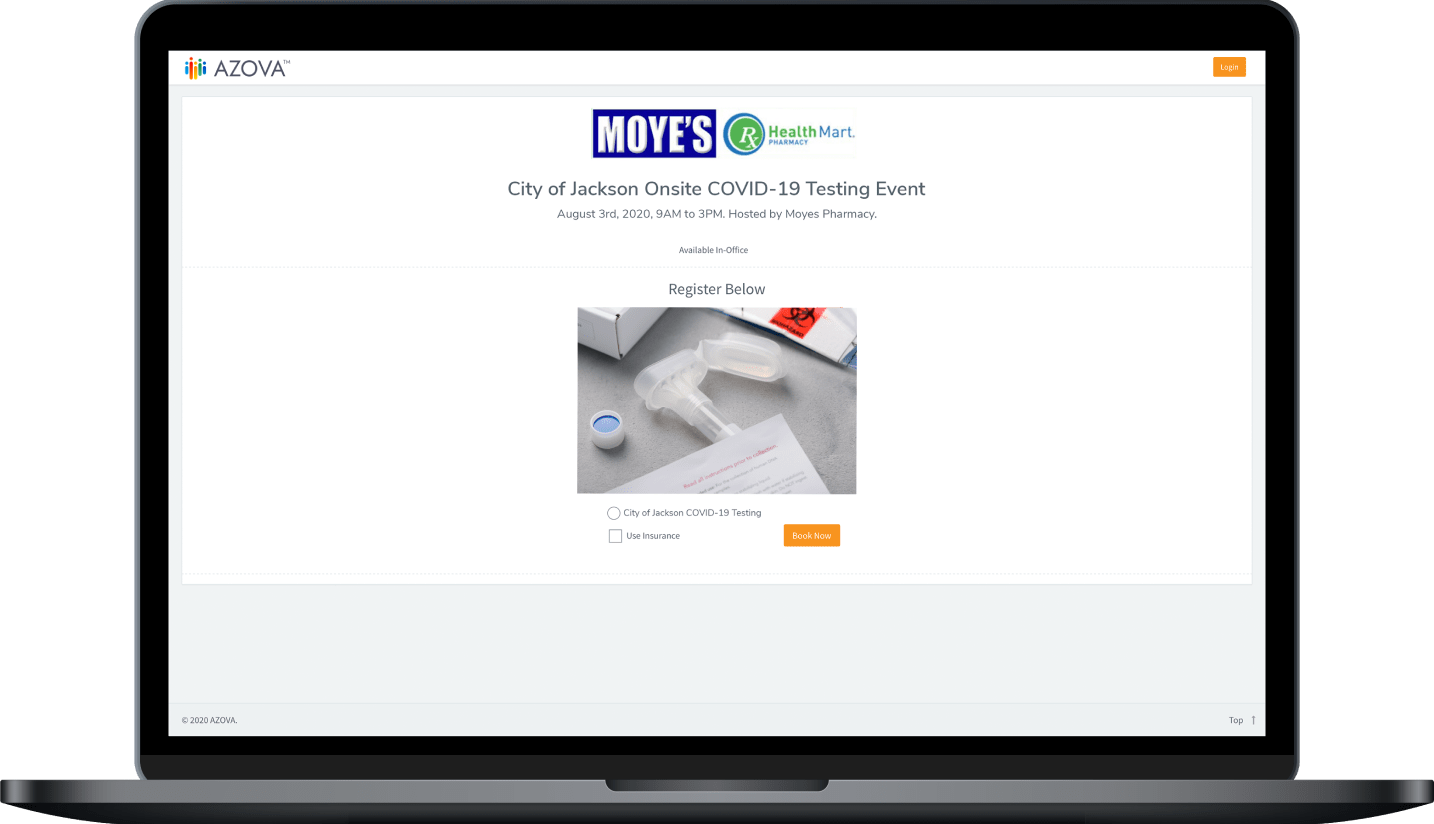
Choose to bill insurance
When you join the national AZOVA COVID testing network, you will receive a custom registration link to use for people who want to use their insurance. Provide this link via email, text or post it on a sign at your pharmacy(s). Have the patient register online through this link. Once registered, you will order the electronic lab order and provide the testing kit to the patient. The patient provides a saliva sample and can drop the kit box in the mail or you can drop the kit box in the mail. Electronic results will be returned to you and the patient simultaneously.
Important note about commercial payers!
Some pharmacies are getting paid by some commercial carriers for providing COVID testing to schools and employers through AZOVA This is a very exciting opportunity for pharmacies. Please check with each carrier and verify that they will pay you and which codes you must submit in order to get paid before you consent to manage a testing event for an organization or you can charge a cash rate for each test to the organization.
Medicare
As a pharmacist, you can apply for an emergency Medicare ID. Approval can be as fast as two days. Once approved, you can submit a claim to medicare for any testing you are doing. Pharmacies can submit a claim to Medicare for COVID testing. This test reimburses at $100 per test and then an administration fee that is approximately $23.
Please ask us about our medical billing service for pharmacies who need medical claims submission services.
About the test
- This test has an FDA authorized Emergency Use Authorization that allows for self-administration or administration by a healthcare provider.
- The device has a 30 month shelf life.
- This test has very high sensitivity and specificity rates.
| Assay | Sensitivity | Specificity | False Positive Rate | False Negative Rate |
|---|---|---|---|---|
| COVID-19 Saliva PCR | 98% | 99% | <1% | 2% |
| COVID-19 Swab PCR | 96% | 97% | <1% | 2% |
Cost per test
Test kit pricing is based upon availability.
Retail single use kits: Sold in minimum quantities of 30, and 60 single use kits.
Exclusive pricing is available to members of the Digital Pharmacy Clinical Services Network. Please ask your sales rep for details.
Onsite event bulk test kits: Sold in minimum quantities of 200 kits.
Exclusive pricing is available when you organize on site testing events. This program is available only to members of the Digital Pharmacy Clinical Services Network.
Join the network and your network manager will contact you with pricing and availability.
To participate in the program, you must join the AZOVA Digital Clinical Services Network. Membership in the network is $1/month per pharmacy location. All software that you will need is provided in your network membership.
It will take 20 minutes to onboard if you have everything ready before you start the sign up process.
The pharmacist in charge for your pharmacy should complete the onboarding process. If you have more than one location, choose the pharmacist in charge at one location to complete the onboarding process for all locations. This pharmacist will become the admin for your business on AZOVA and can add as many locations as needed. Add the email address of the PIC at each location during your onboarding.
You will need your personal pharmacist NPI number, each pharmacy’s NPI number, and the ACH information for your company (this information will be used to pay you). Note: Your membership also includes video calling, SMS notifications and FREE secure messaging for your pharmacist.
You will be asked to enter your credit card during the onboarding process. Your card will be charged the $1 membership fee and, for SMS and video calling (if you choose to use them). If you do not use the SMS and video calling, your card will not be charged. Video calling is $.03/minute. SMS is $.02/SMS. SMS is used only for notifications when you have a new appointment, an incoming video call, or an incoming message. You can choose to turn it on or off. Secure messaging is FREE OF CHARGE and unlimited unless you choose to upgrade to AZOVA's web chat tool.
Five simple steps and you’re ready to go
Step 1: When you onboard to the network, you will click to participate in the COVID-19 testing program. This will list your pharmacy on the COVID-19 testing pharmacy locator and scheduling tool so patients can find and schedule for your testing services. Patients must register online for all testing. You will also use AZOVA to electronically submit all lab orders
Step 2: You can choose to offer any or all of the following types of COVID-19 tests (as new tests become available, you can choose to offer those as well):
- Self-administered saliva or nasal swab COVID-PCR test. Patients purchase the kit from your pharmacy and can self test at home and drop the kit in the mail or can self-test at your pharmacy.
- More exciting testing technologies are on the way so stay tuned!
Step 3: You can order all testing supplies through AZOVA.
Step 4: Electronically submit each lab test order to the lab through the AZOVA electronic health record system. AZOVA is interfaced with the testing laboratories. Your laboratory orders will be transmitted electronically and results are reported directly to you and your patients.
Step 5: AZOVA reimburses you what you paid for the test kit plus $22 to $29 for each test that you administer to patients who register through AZOVA for an in pharmacy test.
Once you’ve finished your pharmacy sign up, you are required to complete an online training. Once you are certified through AZOVA, you will be listed in the testing network and can begin testing.

